What is Lotrell Ophthalmic Solution?
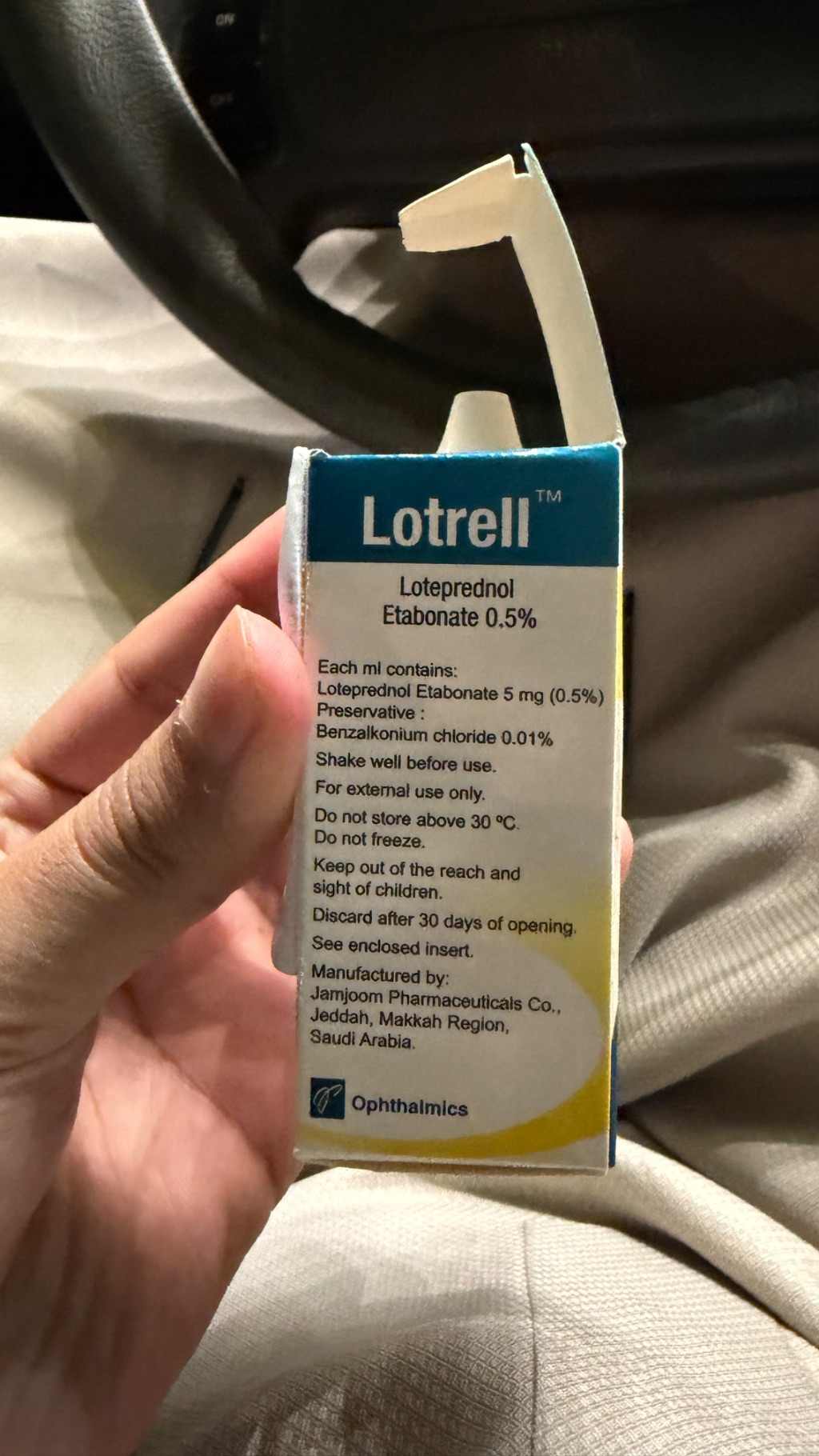
Lotrell Ophthalmic Solution is a prescription-strength eye drop containing the corticosteroid Loteprednol Etabonate. It is specifically formulated to treat inflammation and swelling in the eyes, often prescribed after eye surgery or for conditions like allergic conjunctivitis or uveitis. As a potent anti-inflammatory agent, it is not available over-the-counter and requires close medical supervision due to its powerful effects and potential side effects. This solution falls into the category of ophthalmic steroids, which are distinct from general lubricating eye drops or allergy relief products.
The ingredient breakdown
This prescription ophthalmic solution features a focused ingredient list, primarily centered around its active pharmaceutical component and a common preservative. While designed for targeted efficacy, understanding each ingredient's role and potential impact is crucial for informed use, especially given its potent nature.
The primary active ingredient in Lotrell Ophthalmic Solution is Loteprednol Etabonate, a corticosteroid. Corticosteroids work by suppressing the body's immune response, thereby reducing inflammation, redness, and swelling in the eye. While highly effective for acute inflammatory conditions, prolonged use of potent corticosteroids like Loteprednol Etabonate carries significant risks. The FDA and medical guidelines emphasize that misuse or extended application can lead to serious ocular complications, including increased intraocular pressure (which can progress to glaucoma), cataract formation, and an elevated risk of secondary ocular infections by suppressing the eye's natural defenses.
Another notable ingredient is Benzalkonium chloride (BAK), which serves as a preservative in the solution. BAK is widely used in ophthalmic products to prevent bacterial contamination and maintain sterility once the bottle is opened. However, research indicates that chronic exposure to BAK, particularly in individuals with pre-existing dry eye or glaucoma, can cause ocular surface irritation, damage to corneal and conjunctival cells, and disrupt the delicate balance of the ocular microbiome. While effective at preserving the product, its presence is a factor for consideration with long-term or frequent use, as noted by organizations like the European Medicines Agency (EMA) in their discussions on ophthalmic product excipients.
What this means for your health
Given that Lotrell Ophthalmic Solution is a potent prescription corticosteroid, its use demands strict adherence to your ophthalmologist's instructions. The most significant health implications stem from the active ingredient, Loteprednol Etabonate. Populations who should be extra cautious include individuals with a history of glaucoma, cataracts, or those prone to ocular infections, as the steroid can exacerbate these conditions or mask their progression. Pregnant or breastfeeding individuals should discuss the risks and benefits thoroughly with their doctor, as systemic absorption, though typically low, can occur.
The realistic exposure risk for an average consumer using this product as intended—for a limited duration under medical supervision—is generally managed by their physician. However, prolonged or unsupervised use significantly elevates the risk of serious side effects like irreversible vision loss from glaucoma or the need for cataract surgery. The preservative Benzalkonium chloride also poses a risk of ocular surface toxicity and irritation, particularly for individuals with sensitive eyes or those requiring long-term treatment, potentially worsening conditions like dry eye syndrome.
Safer alternatives to consider
For individuals seeking to manage ocular inflammation, discussing various treatment approaches with a healthcare provider is paramount. Safer alternatives, depending on the specific condition and severity, might include non-steroidal anti-inflammatory drugs (NSAIDs) for certain types of inflammation, or preservative-free ophthalmic solutions to minimize ocular surface irritation, especially for chronic conditions. For mild allergies or irritation, over-the-counter lubricating drops or antihistamine drops that do not contain potent corticosteroids or irritating preservatives could be considered. Always prioritize your doctor's recommendation for prescription-strength conditions.
The bottom line
Lotrell Ophthalmic Solution is a highly effective medication for treating significant ocular inflammation, a critical tool when prescribed and monitored by an ophthalmologist. However, its potent corticosteroid, Loteprednol Etabonate, carries substantial risks of increased intraocular pressure, cataracts, and infection with prolonged or unsupervised use. The preservative Benzalkonium chloride also presents concerns for ocular surface health. For the average reader, this product should only be used under strict medical guidance for the shortest duration necessary, never as a self-prescribed remedy, to ensure its benefits outweigh its considerable risks.